
AR-320 (Suvratoxumab)
Fully Human mAb Targets S. aureus Alpha-toxin
Lung colonization by Staphylococcus aureus bacteria is a risk factor for pneumonia disease. Approximately one in four patients who are with S. aureus progress to ventilator associated pneumonia (VAP). Given that antibiotics are not indicated for the prevention of pneumonia, there is a need for an alternative pre-emptive treatment to reduce the risk of pneumonia in S. aureus colonized patients. AR-320 (suvratoxumab) is being evaluated as a pre-emptive treatment in S. aureus colonized, mechanically ventilated patients in the ICU. It is fully human monoclonal IgG1 antibody (mAb) that specifically targets S. aureus alpha-toxin, an important virulence factor that is secreted by both methicillin-resistant S. aureus (MRSA) and methicillin-sensitive S. aureus (MSSA). AR-320 is half-life extended, that is, able to sustain effective toxin neutralizing activities for approximately 3 months post-dose and above baseline level at one-year post-dose.
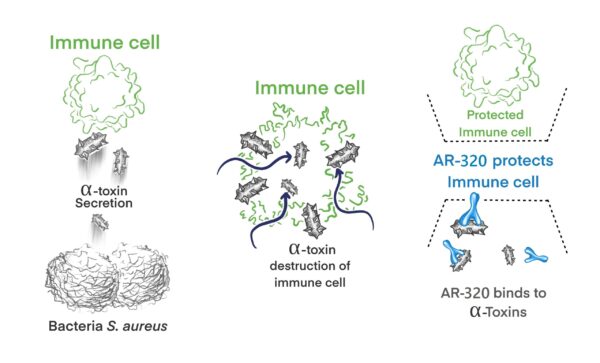
Similar to AR-301’s mechanism of action, AR-320 protects against alpha-toxin mediated destruction of host cells, preserving the human immune cells. AR-320’s mode of action is independent of the antibiotic resistance profile of S. aureus and it is active against infections caused by both MRSA and MSSA.
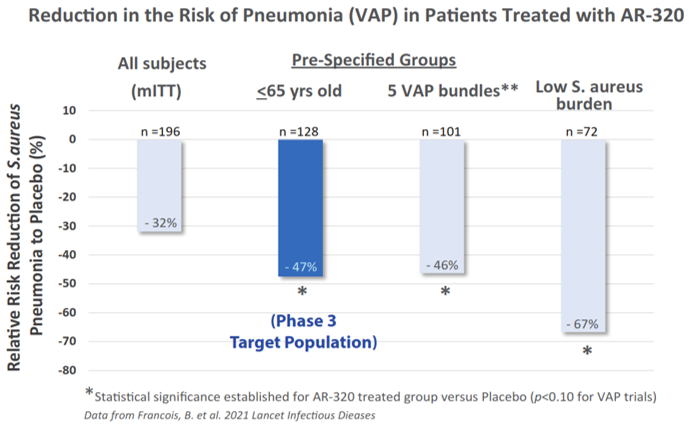
Clinical trial data was recently published in Lancet Infectious Diseases from a double-blinded, placebo-controlled global Phase 2 study conducted across 50 hospital ICUs in Europe and U.S.. The study evaluated the safety, pharmacokinetics, and efficacy of AR-320 in reducing the risk of VAP (ventilator Associated pneumonia) in S. aureus colonized, mechanically ventilated ICU adults (>18 yrs.) who do not have pneumonia. AR-320 was shown to exhibit favorable safety profile and reduced relative risk of S. aureus VAP by 32% in the mITT (microbiological intend-to-treat) population. In a prespecified subpopulation of 65 or younger, AR-320 reduced the risk of VAP by 47%, which reached statistical significance (see figure below). Patients treated with AR-320 consistently demonstrated shorter length of stay in the hospital (~9 days fewer), shorter ICU stay (~3 days), and shorter mechanical ventilation days (~2 days) compared to the placebo group.
AR-320 was granted Fast Track designation by the FDA. Initiation of a global Phase 3 pivotal trial is expected in the 1H2022.
Pathogenicity of S. aureus
S. aureus is a leading cause of hospital-acquired lower respiratory tract infections (e.g., pneumonia), bloodstream, skin, soft tissue, and surgical site infections. Treatment of these infections has become more challenging due to the emergence of multi-drug resistant (‘MRSA’) strains. In many developed countries around the world, including the U.S., UK and Japan, resistance to beta-lactam antibiotics in MRSA strains is a major problem in hospitals and other healthcare settings. Notably, the incidence rate of all invasive MRSA infections, including those outside of hospitals, is high compared with other bacterial pathogens, with these infections resulting in mortality rates of up to 50%.
Despite preventive measures and various antibiotic treatment options, there is a clear medical need for alternative therapeutic interventions to treat hospital-acquired S. aureus infections. In this context, the use of human monoclonal antibodies (mAb) for passive immunotherapy against S. aureus infections represents a promising new approach to combat the disturbing rise in antibiotic resistance.


