
AR-301 (Tosatoxumab)
Fully Human mAb Against Staphylococcus aureus
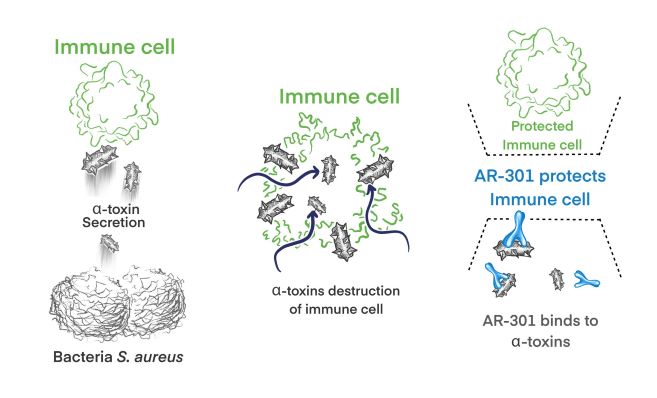
AR-301 (tosatoxumab) is a fully human monoclonal IgG1 antibody (mAb) that specifically targets S. aureus alpha-toxin, an important virulence factor that is secreted by both methicillin-resistant S. aureus (MRSA) and methicillin-sensitive S. aureus (MSSA).
AR-301 protects against alpha-toxin mediated destruction of host cells, preserving the human immune cells. AR-301’s mode of action is independent of the antibiotic resistance profile of S. aureus, and it is active against infections caused by both MRSA and MSSA.
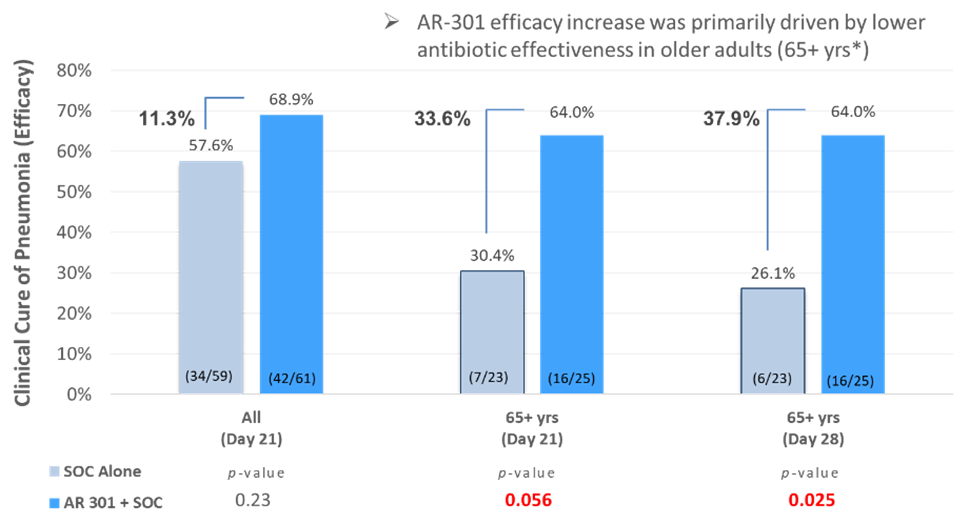
Top-line data was reported from the first of two planned global Phase 3 clinical trials evaluating the safety, pharmacokinetics, and efficacy of a single dose of AR-301 as an adjunctive therapy to standard of care antibiotics in patients diagnosed with ventilator associated pneumonia (VAP) patients caused by S. aureus bacteria(n=174). AR-301 was shown to be well tolerated in the overall full analysis data set ‘FAS’ population. In the microbiologically confirmed full analysis data set ‘mFAS’ population (n=120), a clinically meaningful improvement trend in clinical cure rate at day 21 of 11.3% (p=0.23) was observed.
In the more vulnerable population of older than 65 years of age (n=48), which was a prespecified population in the study, a substantial enhancement in clinical cure rate was observed, by ~3-fold to 33.6% at day 21 (p=0.056) as compared to the improvement that was observed in the overall mFAS, and maintained such increase in clinical cure when assessed at day 28 (37.9%, p=0.025) [see bar graph].
The prespecified healthcare utilization analyses of the mFAS population showed a reduction in the median days on mechanical ventilation, days in the ICU, and days in the hospital of 2, 7, and 14, respectively, when treated with AR-301.
The superior clinical improvement compared to SOC antibiotics alone, especially in the older adults >65 yrs, shows a clear advantage over the available antibiotic therapies. AR-301 showed a substantial and clinically meaningful effect on the underlying S. aureus infection and progression of VABP that is likely to have a sustained disease-modifying effect, especially in the older adult’s population. The safety data suggest that AR-301 also presents an important safety advantage compared to new combinations of antibiotics.
Pathogenicity of S. aureus
S. aureus is a leading cause of hospital-acquired lower respiratory tract infections (e.g., pneumonia), bloodstream, skin, soft tissue, and surgical site infections. Treatment of these infections has become more challenging due to the emergence of multi-drug resistant (‘MRSA’) strains. In many developed countries around the world, including the U.S., UK, and Japan, resistance to beta-lactam antibiotics in MRSA strains is a major problem in hospitals and other healthcare settings. Notably, the incidence rate of all invasive MRSA infections, including those outside of hospitals, is high compared with other bacterial pathogens, with these infections resulting in mortality rates of up to 50%.
In spite of preventive measures and various antibiotic treatment options, there is a clear medical need for alternative therapeutic interventions to treat hospital-acquired S. aureus infections. In this context, the use of human monoclonal antibodies (mAb) for passive immunotherapy against S. aureus infections represents a promising new approach to combat the disturbing rise in antibiotic resistance.


